Whey protein is a heterogeneous mixture of proteins. Whey proteins are made up of different protein fractions. Cow (Bos taurus) milk contains 3.5% protein, of which 80% is casein and 20% whey protein. The biological value (BV) of whey protein is 104, while that of casein is 77. However, the BV of total milk protein is 91[33]. BV is a coefficient that shows how much of the protein absorbed in the body's intestinal tract is consumed for the creation of new proteins, but not for energy needs [34].
Most whey proteins are compactly coiled globular molecules that are mainly hydrophobic and linked by peptide bonds [30].
Properties of whey protein:
- helps to stimulate appetite suppression and satiety [8,9,10, 11];
- is a high-quality protein source rich in all essential amino acids and other biologically active substances that help reduce fat mass and preserve muscle mass, as part of a complete balanced diet and physical activity [13,14,15,16];
- helps maintain a positive level of glutathione by providing the necessary amino acids (glutamic acid, cysteine, glycine) in the diet, also by modeling their synthesis [17,18];
- facilitates the absorption of dietary minerals, vitamins and fatty acids [17,20,21];
- helps improve mood and concentration [17,20,21];
- helps preserve and reduce body protein loss (muscle) due to sacropenia in older people [12,22,23,24];
- are easier to digest and absorb compared to other protein sources [17,21,21].
- whey proteins have 10-30% more "value" than barley, rice, soy and pea proteins, according to the Digestible Indispensable Amino Acid Score (DIAAS) method of the International Food and Agriculture Organization (FAO) [ 25]
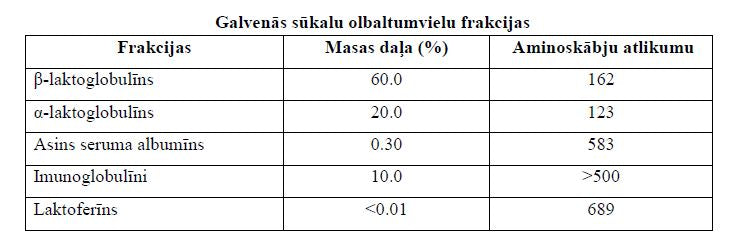
- is a globular protein of 162 amino acid residues [30];
- is very rich in branched chain amino acids (25.1%), especially leucine (13.5%) [35];
- is synthesized in the cow's mammary gland [35];
- contains two –S-S- bonds and one free thionyl group [30];
- constitutes 50-60% of whey proteins and 10% of total milk proteins (2-4 g/l) [35].
- α-Lactalbumin is a monomeric globular calcium-containing protein composed of 123 amino acid residues, including lysine, leucine, threonine, tryptophan, and cysteine [36];
- contains four –S-S- bonds [30];
- is synthesized in cow mammary gland cells and acts as a regulatory component in the enzymatic system and is responsible for the biosynthesis of lactose [36];
- constitutes 20% of whey proteins and 3.4% of total milk proteins (0.6-1.7 g/l) [36];
In contrast to β-lactoglobulin, α-lactoalbumin has a very low immunogenicity, thus a low allergy-inducing potential, which makes it a suitable nutrient for milk-allergic children [ 21 ].
The health-promoting effects of α-lactoalbumin are divided into three categories:
- the effect of whole intact protein;
- effects of individual amino acids from digested protein;
- effect formed by peptides in the gastrointestinal tract by hydrolyzing proteins.
- is a monomeric, globular Fe3+-binding glycoprotein of 689 amino acid residues [39];
- enters milk from blood plasma [39];
- belongs to the group of transferrins, which are blood plasma metal-binding and transporting proteins [39];
- lactoferrin constitutes 1% of whey protein [39];
- lactoferrin has two Fe3+ binding sites. Each of them can bind 1.4 mg Fe3+ /g [40].
Lactoferrin is also used in toothpastes and mouthwashes, where in complex with lactoperoxidase and lysozyme it performs an antibacterial function [28].
- constitutes 3% of whey proteins [38];
- is a globular protein consisting of 583 amino acid residues [38];
- it is not synthesized in the mammary gland, but enters the milk from the blood plasma [38];
- contains 17 -S-S- bonds [30].
- similar to β-lactoglobulin, binds hydrophobic molecules incl. fatty acids [38];
- blood serum albumin contains all essential amino acids [38].
- also called "antibodies", make up 9-10% of whey proteins [20];
- are glycoproteins composed of four peptide chains linked by disulfide bonds [41];
- nutritionally, it is a good source of the essential amino acid, cysteine [41].
Amino acids in whey proteins are found in the fractions described above and, minimally, also in the form of free amino acids. The amino acid content of whey proteins can vary and can be influenced by various factors [38]. Whey proteins contain all essential amino acids, they make up 60% of the total amino acids in them [30].
Whey proteins are an excellent source of sulfur-containing (methionine, cysteine) amino acids. The ability of sulfur-containing amino acids to promote immune function and the body's antioxidative capacity by modeling glutathione synthesis is of great importance in the body [28]. Glutathione is an endogenous antioxidant present in every cell of the human body. Especially in the liver, where it provides detoxification of cells and the lymphatic system. A positive amount of glutathione stimulates the functioning of the immune system, it is directly related to the aging processes of the body. Glutathione biosynthesis in the body requires glutamic acid, cysteine and glycine [42].
Whey proteins are also a dietary source of branched chain amino acids (isoleucine, leucine, valine). Branched chain amino acids play an important role in skeletal muscle protein synthesis, and they can also be involved in the energetic metabolism of muscle tissue [28].
After whey OBV is digested in the gastrointestinal tract, not all amino acids participate in the energy metabolism of the human body and the formation of plastic material. Some of the proteins are enzymatically cleaved to peptides of different lengths, in which the number of monomers can vary from 2 to 20 amino acids. These discrete amino acid sequences are inactive in the protein from which they came. In their free form, they have a wide range of biological activity, including antimicrobial, antihypertensive, and immunomodulating activity[43].

9. Anderson, H., Moore, S. Dietary proteins in the regulation of food intake and body weight in humans. Journal of Nutrition, 2004, Vol. 134, Iss. 4, p. 974-979.
10. Hall, W. L., Millward, D. J., Long, S. J., Morgan, L. M. Casein and whey exert different effects on plasma amino acid profiles, gastrointestinal hormone secretion and appetite. British Journal of Nutrition, 2003, Vol. 89, Iss. 2, p. 239-248.
12. Ha, E., Zemel, M. Functional properties of whey, whey components and essential amino acids: mechanism underlying health benefits for active people. Journal of Nutritional Biochemistry, 2003, Vol. 14, Iss. 5, p. 251-258.
42
13. Layman, D., Baum, J. Dietary protein impact on glycemic control during weight loss. Journal of Nutrition, 2004, Vol. 134, Iss. 4, p. 968-973.
14. Layman, D. K., Boileau, R. A., Erickson, D. J., Painter, J. E., Shiue, H., Sather, C., Christou, D. D. A reduced ratio of dietary carbohydrate to protein improves body composition and blood lipid profiles during weight loss in adult women. Journal of Nutrition, 2003, Vol. 133, Iss. 2, p. 411-417.
15. Layman, D. K., Shiue, H., Sather, C., Erickson, D. J., Baum, J. Increased dietary protein modifies glucose and insulin homeostasis in adult women during weight loss. Journal of Nutrition, 2003, Vol. 133, Iss. 2, p. 405-410.
16. World Health Organization, Food and Agricultural Organization of the United Nations, Vitamin and mineral requirements in human nutrition, second edition, 2004.
18. Middleton, N., Jelen, P., Bell, G. Whole blood and mononuclear cell glutathione response to dietary whey protein supplementation in sedentary and trained male human subjects. International journal of food sciences and nutrition, 2004, Vol. 55, Iss. 2, p. 131-141.
19. Zavorsky, G. S., Kubow, S., Gray, V., Riverin, V., Lands, L. C. An open-label dose-response study of lymphocyte glutathione levels in healthy men and women receiving pressurized whey protein isolate supplements. International journal of food sciences and nutrition, 2007, Vol. 58, Iss. 6, p. 429-436.
20. Graham Knowles and Harsharnjit S. Immunomodulation by Dairy Ingredients: Potential for Improving Health. In: Gill Colette Shortt, John O'Brien. Handbook of functional dairy products. 2004, p. 128-133.
21. A. Kanekanian. Milk and dairy products as functional foods. In: Proteins in Human Health, Casein and Whey. Wiley. 2014, c. 4, p. 94-146.



1 comment
Labdien. Kādas ir jūsu domas par to, ka kazeīns veicina vēža augšanu (no grāmatas “lielais Ķīnas pētījums”. Īsumā – veica petijumus, kur pelēm un žurkām deva kancorogenas vielas un žurkām/pelēm, kuram uztura nebija kazeins vēzis neveidojas , bet kur bija kazeins vēzis veidojas. Paldies